Tandem Diabetes Care ProductsCreating New Possibilities for People Living with Diabetes
We know that diabetes management is a personal journey to keep blood glucose balanced throughout the day and night. That’s why we deliver innovative insulin pumps, automated insulin delivery systems, supplies, and technology.
Choose a Pump
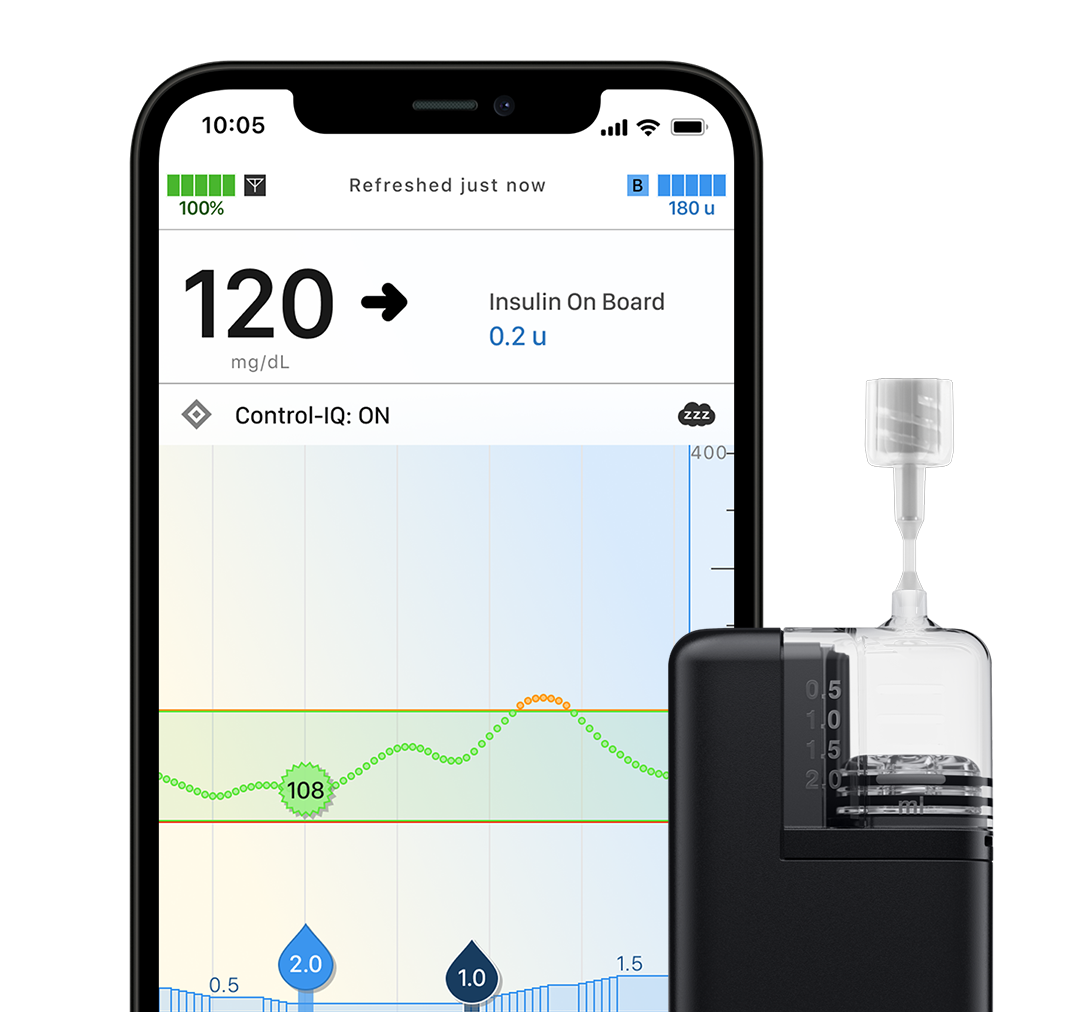
Tandem Mobi System
Impressively small and can be worn almost anywhere,* Tandem Mobi gives you more options for how you manage your diabetes.
Available Features
![]() Powered by Control-IQ+ technology
Powered by Control-IQ+ technology
![]() Capable of remote software updates†
Capable of remote software updates†
![]() Smartphone control (iOS only)‡
Smartphone control (iOS only)‡
![]() Holds 200 units of insulin
Holds 200 units of insulin
![]() Pump button for bolus delivery
Pump button for bolus delivery
![]() Dexcom G6 and G7 sensor compatibility§
Dexcom G6 and G7 sensor compatibility§
![]() Inductive wireless charging
Inductive wireless charging
![]() On-body wear with adhesive sleeve^
On-body wear with adhesive sleeve^
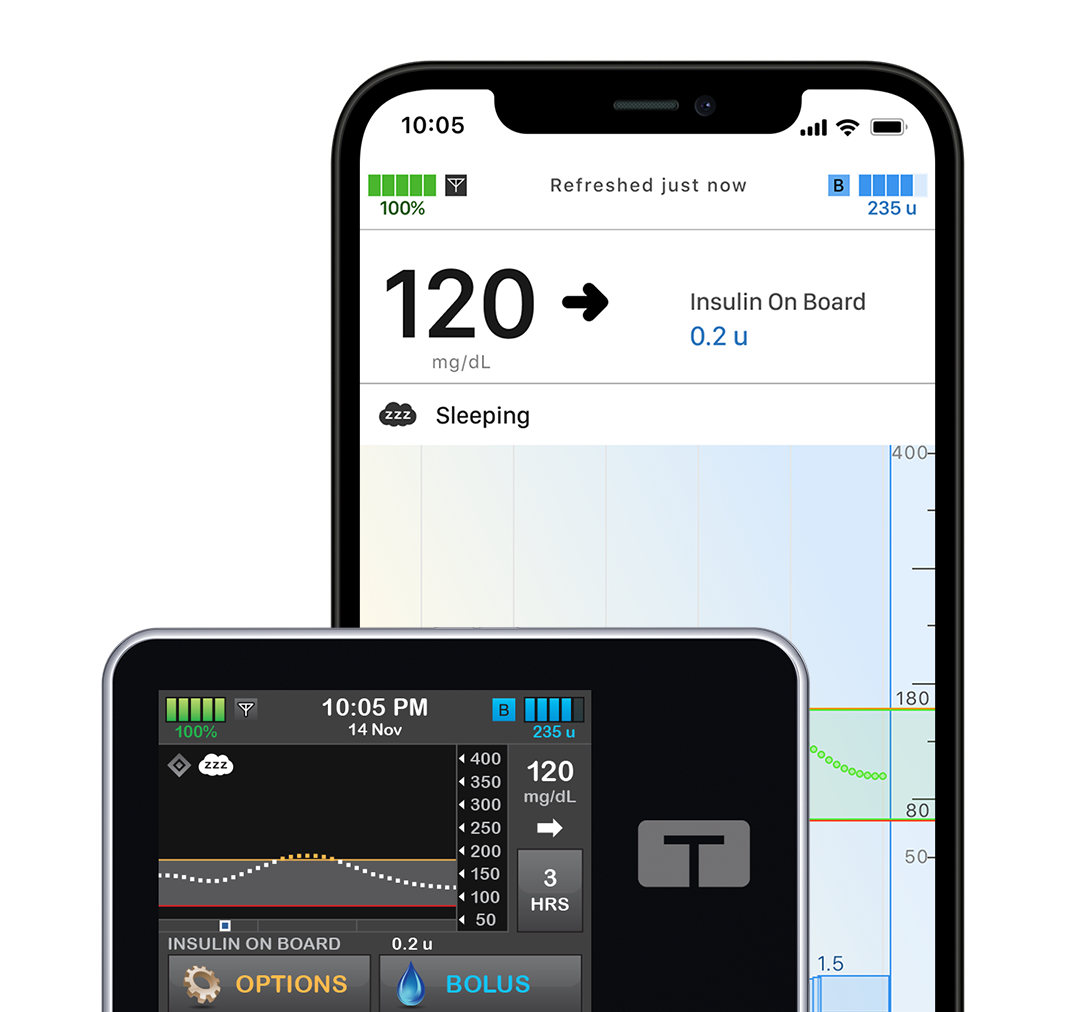
t:slim X2 Insulin Pump
With a sleek color touchscreen, this all-in-one insulin pump integrates seamlessly into your life like a sophisticated, modern device.
Available Features
![]() Powered by Control-IQ+ technology
Powered by Control-IQ+ technology
![]() Capable of remote software updates†
Capable of remote software updates†
![]() Mobile bolus delivery (iOS & Android)‡
Mobile bolus delivery (iOS & Android)‡
![]() Holds 300 units of insulin
Holds 300 units of insulin
![]() Pump button for bolus delivery
Pump button for bolus delivery
![]() Dexcom G6 and G7 sensor compatibility§
Dexcom G6 and G7 sensor compatibility§
![]() FreeStyle Libre 2 Plus compatibility§
FreeStyle Libre 2 Plus compatibility§
![]() Integrated color touchscreen
Integrated color touchscreen
Get started today!
A Tandem Pump Specialist will help you get started.

Choose an Infusion Set
Disconnect. Reconnect. It’s a Snap!
Finding the right infusion set is an important part of successful insulin pump therapy. Choose from different insertion angles, materials, and a variety of a tubing lengths.


Discover Innovative Tools
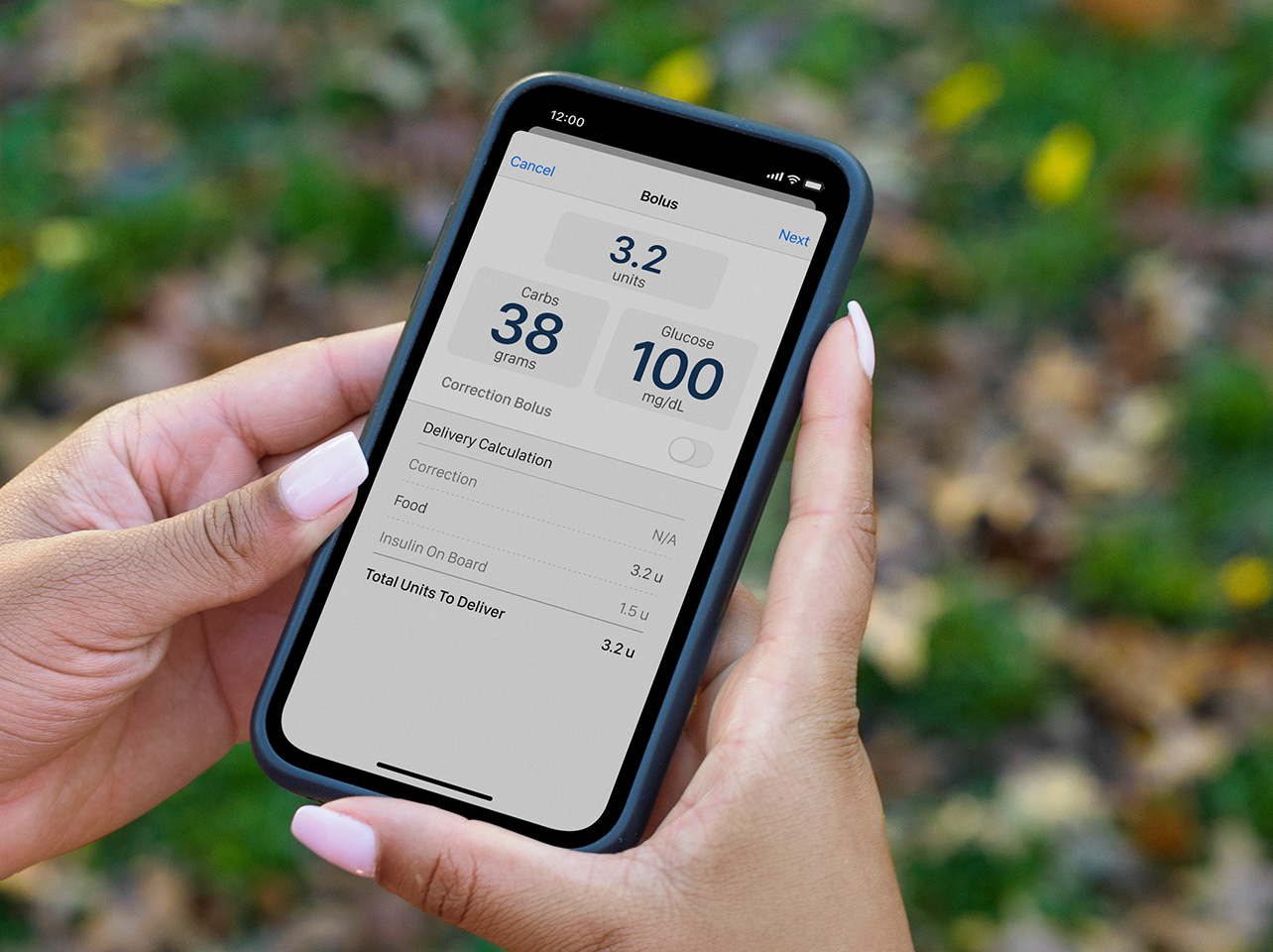
Connected Care System
We are more than an insulin pump company. We also support users through cutting-edge mobile and cloud-based applications that are designed to help you see therapy data and better manage your diabetes.

Choose Pump Accessories
Personalize Your Pump
We know that your insulin pump is an integral part of your life. That’s why we offer a wide range of pump accessories for wearability, power sources, and protection that meet your needs.
