Tandem Diabetes Care ProductsCreating New Possibilities for People Living with Diabetes
We know that diabetes management is a personal journey to keep blood glucose balanced throughout the day and night. That’s why we deliver innovative insulin pumps, automated insulin delivery systems, supplies, and technology.

Compare Pumps

Tandem Mobi System
Impressively small and can be worn almost anywhere,* Tandem Mobi gives you more options for how you manage your diabetes.
Available Features

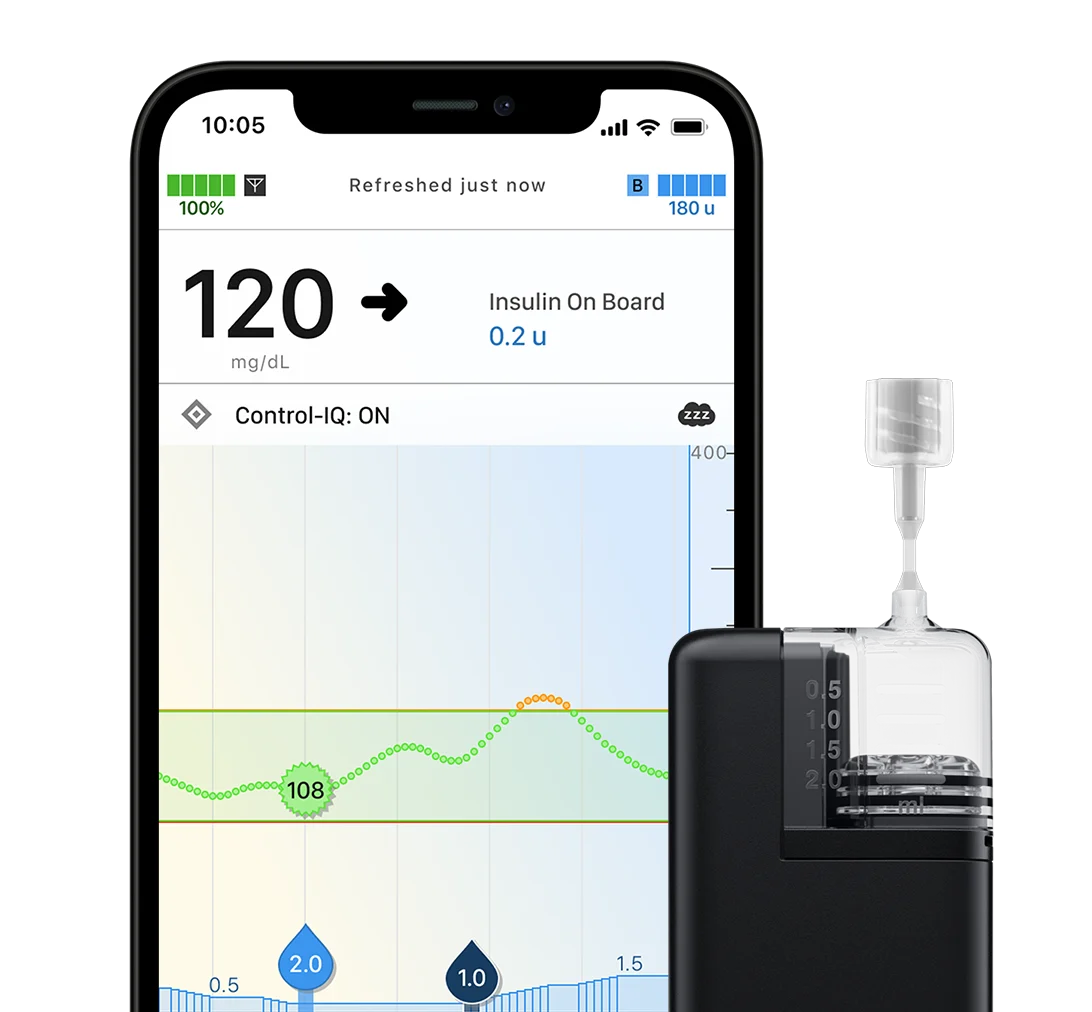
t:slim X2 Insulin Pump
With a sleek color touchscreen, this all-in-one insulin pump integrates seamlessly into your life like a sophisticated, modern device.
Available Features
Order a Tandem Diabetes Care Automated Insulin Delivery System Today.
A Tandem Diabetes Care Specialist will help you get started.

#1 in Overall Satisfaction by Healthcare Providers
You may be able to experience the life-changing benefits of Tandem automated insulin delivery for $0 out of pocket.
Seagrove Partners HCP Perspectives (Devices), September 2025 Insulin Delivery Section N=120.
Cost per day calculation reflects the average monthly out-of-pocket cost for pump and disposables necessary to utilize Tandem pump therapy over the life of the 48-month pump warranty. Assumes consumption of 10 infusion sets and cartridges per month.
$0 out of pocket claim based on direct orders from Tandem customers with commercial insurance from 1/1/2025-12/31/2025. Data on file. Actual cost may vary and is subject to individual insurance coverage.

Choose an Infusion Set
Disconnect. Reconnect. It’s a Snap!
Finding the right infusion set is an important part of successful insulin pump therapy. Choose from different insertion angles, materials, and a variety of a tubing lengths.


Discover Innovative Tools
Connected Care System
We are more than an insulin pump company. We also support users through cutting-edge mobile and cloud-based applications that are designed to help you see therapy data and better manage your diabetes.


Choose Pump Accessories
Personalize Your Pump
We know that your insulin pump is an integral part of your life. That’s why we offer a wide range of pump accessories for wearability, power sources, and protection that meet your needs.

What's next?
Whether you're ready to get a Tandem insulin pump, or looking to check insurance coverage, you’ll find everything you need here.
* Do not wear or place your pump more than 12 inches (30.5 cm) above your infusion site. Doing so may result in over-delivery of insulin.
† Future updates for all or some Tandem products may not be developed and may not be offered everywhere and would be subject to applicable regulatory approvals. Software updates are only available to customers who are in warranty at the time they update their pump. Additional training may be required to access certain software updates. Charges may apply. Tandem may discontinue select software and features over time at its discretion.
‡ Our mobile apps require a compatible smartphone model and operating system (sold separately). For t:slim X2 insulin pump users, bolus delivery also requires an app update, a remote software update on the pump, and additional training.
§ CGM sold separately.
^ Tandem Mobi adhesive sleeve sold separately.
Important Safety Information
RX ONLY.
Indications for Use
Tandem Mobi system: The Tandem Mobi insulin pump with interoperable technology (the pump) is intended for the subcutaneous delivery of insulin, at set and variable rates, for the management of diabetes mellitus in persons requiring insulin. The Pump is able to reliably and securely communicate with compatible, digitally connected devices, including automated insulin dosing software, to receive, execute, and confirm commands from these devices. The pump is indicated for use in persons 2 years of age and greater.
t:slim X2 insulin pump: The t:slim X2 insulin pump with interoperable technology (the pump) is intended for the subcutaneous delivery of insulin, at set and variable rates, for the management of diabetes mellitus in persons requiring insulin. The pump is able to reliably and securely communicate with compatible, digitally connected devices, including automated insulin dosing software, to receive, execute, and confirm commands from these devices. The pump is indicated for use in persons 2 years of age and greater.
Control-IQ+ technology: Control-IQ+ technology is intended for use with compatible integrated continuous glucose monitors (iCGM, sold separately) and alternate controller enabled (ACE) pumps to automatically increase, decrease, and suspend delivery of basal insulin based on iCGM readings and predicted glucose values. It can also deliver correction boluses when the glucose value is predicted to exceed a predefined threshold. Control-IQ+ technology is intended for the management of type 1 diabetes mellitus in persons 2 years of age and greater and of type 2 diabetes mellitus in persons 18 years of age and greater.
Warning: Control-IQ+ technology should not be used in anyone under the age of 2 years old with type 1 diabetes or under the age of 18 years old with type 2 diabetes. It should also not be used in patients who require less than a total daily insulin dose of 5 units of insulin per day or who weigh less than 20 pounds (9 kilograms), as those are the required minimum values needed for Control-IQ+ to operate safely.
Users of the pump and Control-IQ+ must: use the insulin pump, iCGM, and all other system components in accordance with their respective instructions for use. Failure to follow these instructions for use could result in an over delivery or under delivery of insulin. This can cause hypoglycemia (low BG) or hyperglycemia (high BG) events. Visit tandemdiabetes.com/safetyinfo for additional important safety information.
Infusion sets are indicated for the subcutaneous infusion of insulin administered by Tandem insulin pumps for the treatment of diabetes. These infusion sets are indicated for single use. For contraindications, warnings, precautions, and other important information, please refer to the instructions for use accompanying the infusion sets.
Tandem t:slim mobile app: The feature set available within the Tandem t:slim mobile app is dependent on the pump software version and the compatible smartphone’s model and operating system (OS).
The Display and Data Upload feature set provides a secondary display of pump and continuous glucose monitoring (CGM) information, including display of your pump alerts and alarms, and enables wireless uploading of pump and CGM data to the Tandem cloud through an internet or wireless data connection. Standard carrier data rates may apply.
The Bolus Delivery plus Display and Data Upload feature set additionally allows users to request, stop, and cancel a bolus on the pump from the Tandem t:slim mobile app. At least one smartphone security setting must be enabled to utilize the Bolus Delivery feature of the Tandem t:slim mobile app.
WARNING: Always rely on your pump to make therapy decisions when using a smartphone that is incompatible with the Bolus Delivery feature.
PRECAUTIONS: Always rely on your pump to make therapy decisions if using a smartphone that is incompatible, the smartphone is lost or damaged, or the smartphone loses Bluetooth® connectivity with your pump. Important pump alerts and alarms are only received as app notifications when enabled and the app is either active or running in the background.
FreeStyle Libre 3 Plus sensor: For FreeStyle Libre 3 Plus sensor safety information, please visit freestyle.abbott/us-en/safety-information. The sensor housing, FreeStyle, Libre, and related brand marks are marks of Abbott and used with permission.