Note: This post was updated 3/11/2020 to reflect updated messaging regarding Medicare coverage.
We’re excited to announce FDA approval of the t:slim X2™ Insulin Pump with Basal-IQ Technology,
a predictive low glucose suspend feature designed to help reduce the frequency and duration of low blood glucose events (hypoglycemia) using best-in-class glucose sensor data. We’re here to answer some frequently asked questions about our announcement.
Please note these answers are intended for our customers in the United States.
How Does Basal IQ Technology Work?
Integrated with the Dexcom® G6 Continuous Glucose Monitoring (CGM) System, the Basal-IQ feature helps reduce the frequency and duration of low-glucose events by predicting glucose levels 30 minutes ahead
and suspending insulin if they are expected to drop below 80 mg/dL. Insulin delivery resumes once glucose begins to rise. We have a few more details for you here: www.tandemdiabetes.com/products/t-slim-x2-insulin-pump.
What’s so great about Basal-IQ Technology?
A pivotal study showed the use of Basal-IQ Technology on the t:slim X2 Insulin Pump demonstrated a 31% relative reduction in time spent below 70 mg/dL when compared with a CGM-enabled pump without
Basal-IQ Technology. Importantly, this marked reduction of time spent in low glucose was accomplished without any increase in the rate of hyperglycemia. Participants in the study overwhelmingly described the system as simple to learn and use.
1 It is also approved for use by children as young as 6 years old.
"Being a person with diabetes complicates all aspects of my life. The t:slim X2 with Basal-IQ eased some of these burdens. During my time in the Basal-IQ PROLOG study, the feature shined overnight when I could see it suspended and protected me from lows." - Elena E., Clinical Study Subject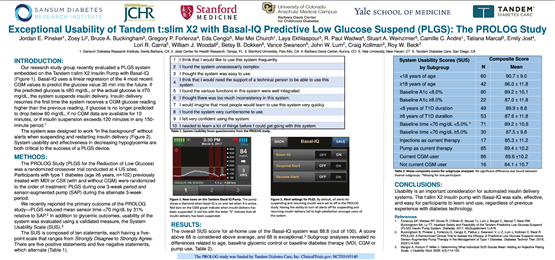
Pinsker, et al. Exceptional Usability of Tandem t:slim X2 Insulin Pump with Basal-IQ Predictive Low Glucose Suspend (PLGS): The PROLOG Study. Poster presented at: American Diabetes Association 78th Scientific Sessions. June 22-26, 2018. Orlando, FL.
"My 13-year-old son manages his care quite well, but his schedule with gym class after lunch often causes his BG to drop. When this happens, his class time is interrupted. With Basal-IQ during the PROLOG study, Russell spent more time in class and less time taking care of his medical needs." - Kathleen B., Mother of Clinical Study Subject
When will Basal IQ Technology be available?
On August 16, we announced we began sending emails with update instructions to in-warranty t:slim X2 Pump users in the United States, who now have the option to add Basal-IQ Technology free
of charge via remote software update. Emails contain the link to the new patient portal and instructions about the process, and will be sent in batches. We anticipate that all emails will go out by the end of August. If you customers can't
wait for an email, they can learn more about the process and how to get started here. (Just know that all will still get the email later, even if they start the process sooner.) https://www.tandemdiabetes.com/products/x2-update
Will Basal IQ Technology work with the Dexcom® G5 Mobile CGM System?
The t:slim X2 Pump with Basal-IQ Technology is only compatible with the latest Dexcom G6 CGM, allowing the system to predict and help prevent lows with zero fingersticks.
*
What does iCGM mean?
Essentially, it’s a faster pathway to deliver new CGM technology. iCGM classification designates approval for integration with other medical devices, such as insulin pumps, based on a series of special control
requirements. Dexcom G6 was the first CGM device to receive iCGM classification from the FDA earlier this year, and Tandem is the first company with a pump approved with iCGM compatibility. The classification of the Dexcom G6 CGM sensor, combined
with our Tandem Device Updater, allows us to offer our Basal-IQ Technology with Dexcom G6 CGM integration without a new submission to the FDA. We are excited about the potential for an exciting new wave of future innovation for the diabetes
community.
Oh no, I just ordered Dexcom G5 Mobile CGM Sensors!
We realize some customers will be affected by the timing of their CGM sensor orders, and we are exploring options for customers who will have Dexcom G5 Mobile CGM supplies when the
software update rolls out. We’d like to note that eligible t:slim X2 Insulin Pump users will be able to take advantage of the Basal-IQ Technology software update, free of charge for some time. The no-cost eligibility is determined by
our release date in 2018 – not your download date – so it’s possible to align your update with your next sensor order.
I have Medicare, can I use Basal-IQ Technology?
Our warranty doesn't prevent delivery of software updates to people insured under government insurance policies. The determination of eligibility for new features by these plans will
depend on whether a prescription is required, and if the new feature falls within other devices that they cover. For example, a Medicare patient is not excluded from being able to download a software update, but Medicare requirements may prevent
use of a new feature that requires integration with another device that is not covered by Medicare.
I have a regular t:slim® Insulin Pump or t:slim G4™ Insulin Pump, how can I get Basal-IQ Technology?
We offered an upgrade program for t:slim and t:slim G4 Insulin Pump users to access the updatable t:slim X2 Insulin Pump
from October 2016–December 2017. Currently, we have no plans to have another upgrade program. If anything changes in your insurance coverage during your warranty period, please let us know. Our insurance verification team can look into
your options. They can be reached at (877) 801-6901, Monday-Friday between 6 AM and 5 PM PT.
What about Canadians? We’re waiting for a new option up here!
It is too early for us to speculate about plans or timing for availability of the t:slim X2 Insulin Pump with Basal-IQ Technology outside the United States. In February,
we announced our submission of a medical device license application to Health Canada to market the t:slim X2 Insulin Pump with Dexcom G5 Mobile CGM integration. We plan to launch that pump in Canada in the second half of 2018, subject to regulatory
approvals.
For additional product and safety information for our customers in the United States, or to begin the order process, visit www.tandemdiabetes.com/tslimX2
or call (877) 801-6901, Monday-Friday between 6 AM and 5 PM PT.
* If glucose alerts and CGM readings do not match symptoms or expectations, use a blood glucose meter to make diabetes treatment decisions.
1. Forlenza GP, Li Z, Buckingham BA, Pinsker JE, et al. Predictive low glucose suspend reduces hypoglycemia in adults, adolescents, and children with type 1 diabetes in an at-home randomized crossover study: Results of the PROLOG trial [published online August 8, 2018]. Diabetes Care. doi:10.2337/dc18-0771.