AutoSoft XC with 5‑inch Tubing
This all-in-one infusion set has just five inches of tubing, which increases the wearability options for the Tandem Mobi system.
6 mm flexible soft cannula
90-degree insertion angle
Automatic insertion device
Rich Text
Quick reversible connector
Easy disconnection
Built-in needle cover
Rich Text
Rich Text
How will you wear Tandem Mobi? This set expands wearability options when paired with the lightweight adhesive sleeve.*
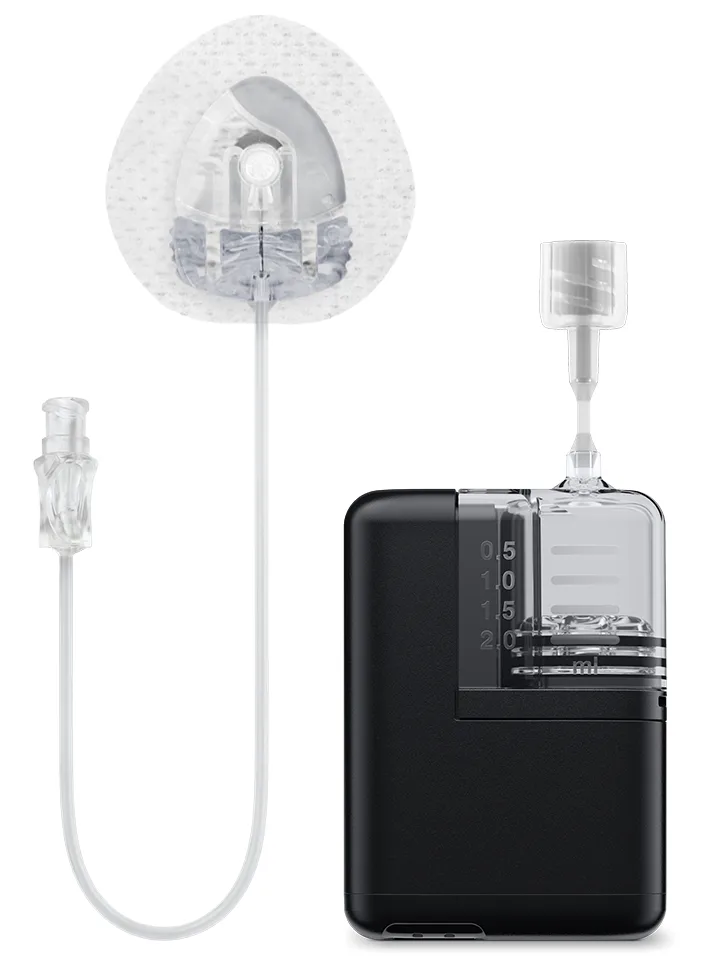
I chose the AutoSoft XC infusion set because of its small size and the multiple placement options it gives me. I also like how each set comes with its own automatic inserter, which helps with smooth application. For me, the insertion is essentially pain-free.
Melody Fox,
Territory Manager at Tandem Diabetes Care

Additional Options
We offer a variety of choices to meet the unique needs of our pump users. Learn more about our other infusion sets and see which one is right for you.

AutoSoft XC
An infusion set with a 90‑degree cannula and large grip for easy disconnection.
Frequently Asked Questions
How do I know if I am eligible for the AutoSoft XC infusion set with 5-inch tubing?
In the Infusion Set Type section of your prescription, your healthcare provider must have specified either Patient Preference or listed AutoSoft XC under Other. To update your prescription, call (877) 801-6901 and select option 3.
How can I order 5-inch tubing?
Infusion sets are ordered in the same way your insulin pump was purchased (i.e., directly from Tandem Diabetes Care or from one of our distributor partners, as determined by your insurance company).
To order supplies directly from Tandem, please call (877) 801-6901 and select option 3. If your insurance allows, you can order different infusion sets and tubing lengths in the same supply order or set up automatic supply reorders. Some distributor partners also offer automatic supply reorders.
How can I order Tandem Mobi adhesive sleeves?
The AutoSoft XC infusion set with 5-inch tubing is designed to be worn with the Tandem Mobi adhesive sleeve. You can purchase more adhesive sleeves through the Store on the Tandem Source platform.
If you are enrolled in our Auto Reorder program and wish to include Tandem Mobi adhesive sleeves to your ongoing orders, please call (877) 801-6901 and select option 3.
* Adhesive sleeve sold separately.
Important Safety Information
RX ONLY.
Indications for Use
Tandem Mobi system: The Tandem Mobi insulin pump with interoperable technology (the pump) is intended for the subcutaneous delivery of insulin, at set and variable rates, for the management of diabetes mellitus in persons requiring insulin. The pump is able to reliably and securely communicate with compatible, digitally connected devices, including automated insulin dosing software, to receive, execute, and confirm commands from these devices. The pump is intended for single patient use. The pump is indicated for use in individuals 2 years of age and greater.
Control-IQ+ technology: Control-IQ+ technology is intended for use with compatible integrated continuous glucose monitors (iCGM, sold separately) and alternate controller enabled (ACE) pumps to automatically increase, decrease, and suspend delivery of basal insulin based on iCGM readings and predicted glucose values. It can also deliver correction boluses when the glucose value is predicted to exceed a predefined threshold. The Tandem Mobi system with Control-IQ+ technology is intended for the management of type 1 diabetes mellitus in persons 2 years of age and greater. Control-IQ+ technology is intended for single patient use. Control-IQ+ technology is indicated for use with NovoLog or Humalog U-100 insulin.
Warning: Control-IQ+ technology should not be used by anyone under the age of 2 years old. It should also not be used in patients who require less than 5 units of insulin per day or who weigh less than 20 pounds.
Control-IQ+ technology is not indicated for use in pregnant women, people on dialysis, or critically ill patients. Do not use Control-IQ+ technology if using hydroxyurea. Users of the Tandem Mobi pump and Control-IQ+ technology must: use the insulin pump, CGM, and all other system components in accordance with their respective instructions for use; test blood glucose levels as recommended by their healthcare provider; maintain sufficient diabetes self-care skills; see healthcare provider(s) regularly; and have adequate vision and/or hearing to recognize all functions of the pump, including alerts, alarms, and reminders. If your CGM readings do not match your symptoms or expectations, use a blood glucose meter to make diabetes treatment decisions. The Tandem Mobi pump, transmitter, and sensor must be removed before MRI, CT, or diathermy treatment. Exposure to MRI, CT, or diathermy treatment can damage the components. Visit tandemdiabetes.com/safetyinfo for additional important safety information.