Time of Initiation of Advanced Hybrid Closed-Loop Therapy and Related Glycemic Outcomes in People with Type 1 Diabetes Transitioning from Multiple Daily Injections
Lars Mueller, Harsimran Singh, Steph Habif, Molly McElwee Malloy, J. Wesley Morberg, Jordan Pinsker, Tandem Diabetes Care
Background
Advanced hybrid closed-loop systems (AHCL) have demonstrated long-term benefits in all people with diabetes (PWD). However, unpublished reports suggest that PWD using multiple daily injections (MDI) might delay activation of AHCL because of concerns around managing pump therapy.
Methods
We retrospectively studied first 90 days of pump activation in MDI users with type 1 diabetes transitioning to t:slim X2 insulin pump with Control-IQ technology. Participants (age 34±20, 52% female) were categorized into 3 groups: G1 initiated Control-IQ technology within 2 days of pump start, G2 initiated between 2-14 days, and G3 initiated within 15-90 days. Hypoglycemia and time in range (TIR) sensor glycemic (SG) metrics for Control-IQ technology use were retrieved from Tandem’s t:connect web application. Group differences were analyzed using Mann Whitney U and Kruskal-Wallis tests.
Results
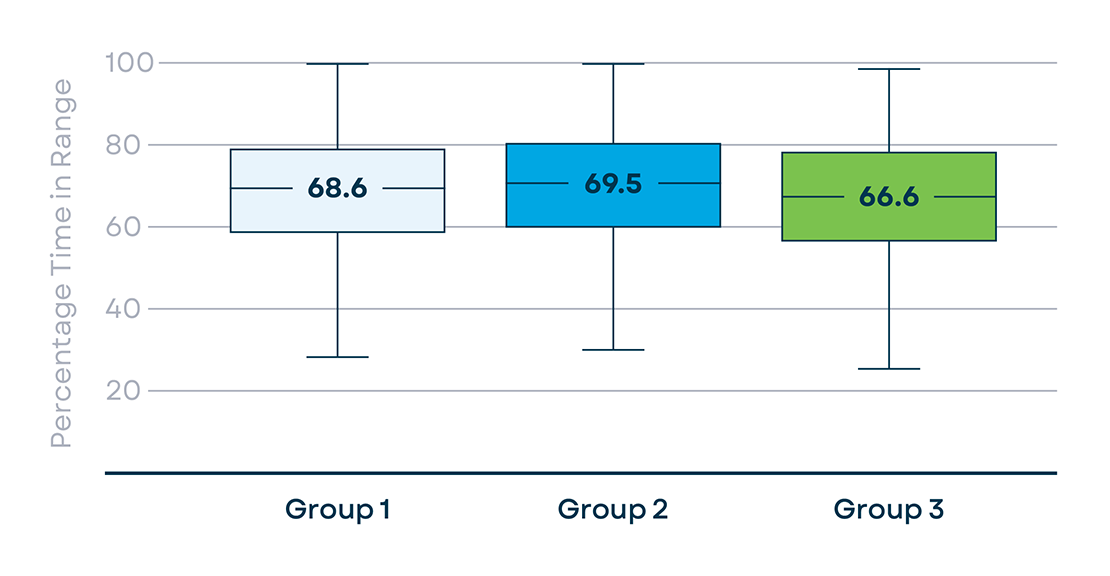
81% of users, initiated Control-IQ technology within 2 days of pump start (G1, n=14222) followed by G2 (14%, n=2448), and G3 (5%, n=870). Groups did not differ on baseline HbA1c. With Control-IQ technology, no differences were noted for SG<54mg/dL (median (IQR): G1=0.13(0.04-0.3), G2=0.12(0.04-0.31), G3=0.13(0.04-0.32) and SG<70mg/dL: G1=0.87(0.36-1.8), G2=0.84(0.38-1.7), G3=0.89(0.37-1.72). There were no differences between G1 and G2 on TIR (70-180mg/dL): G1=68.6(57.8-78.1), G2=69.5(59.1-79.1). However, G3 showed significantly lower TIR: 66.6(55.6-77.9) (p=0.003). Overall, 99.5% of participants continued to use Control-IQ technology at the time of this analysis.
Median Sensor Time in Range (70-180 mg/dL)
Conclusion
95% of MDI users (G1, G2) successfully initiated Control-IQ technology within 14 days of pump start. While all groups experienced success with Control-IQ technology, G3 showed lower TIR with delayed initiation.