Real-World Psychosocial Outcomes and Ease of Use Predict Trust in Automated Insulin Delivery Systems
Michelle Manning,* Molly McElwee-Malloy,* Keaton Stoner,† and Steph Habif*
Objective
To better understand the formation of trust in automated insulin delivery (AID) systems by examining the relationship between trust and psychosocial outcomes and AID system usability.
Method
Individuals with diabetes responded to an online survey administered by dQ&A, which assessed variables of interest, trust, AID system perceived usability, psychosocial outcomes, basic demographics, and information on method of diabetes management.
Results
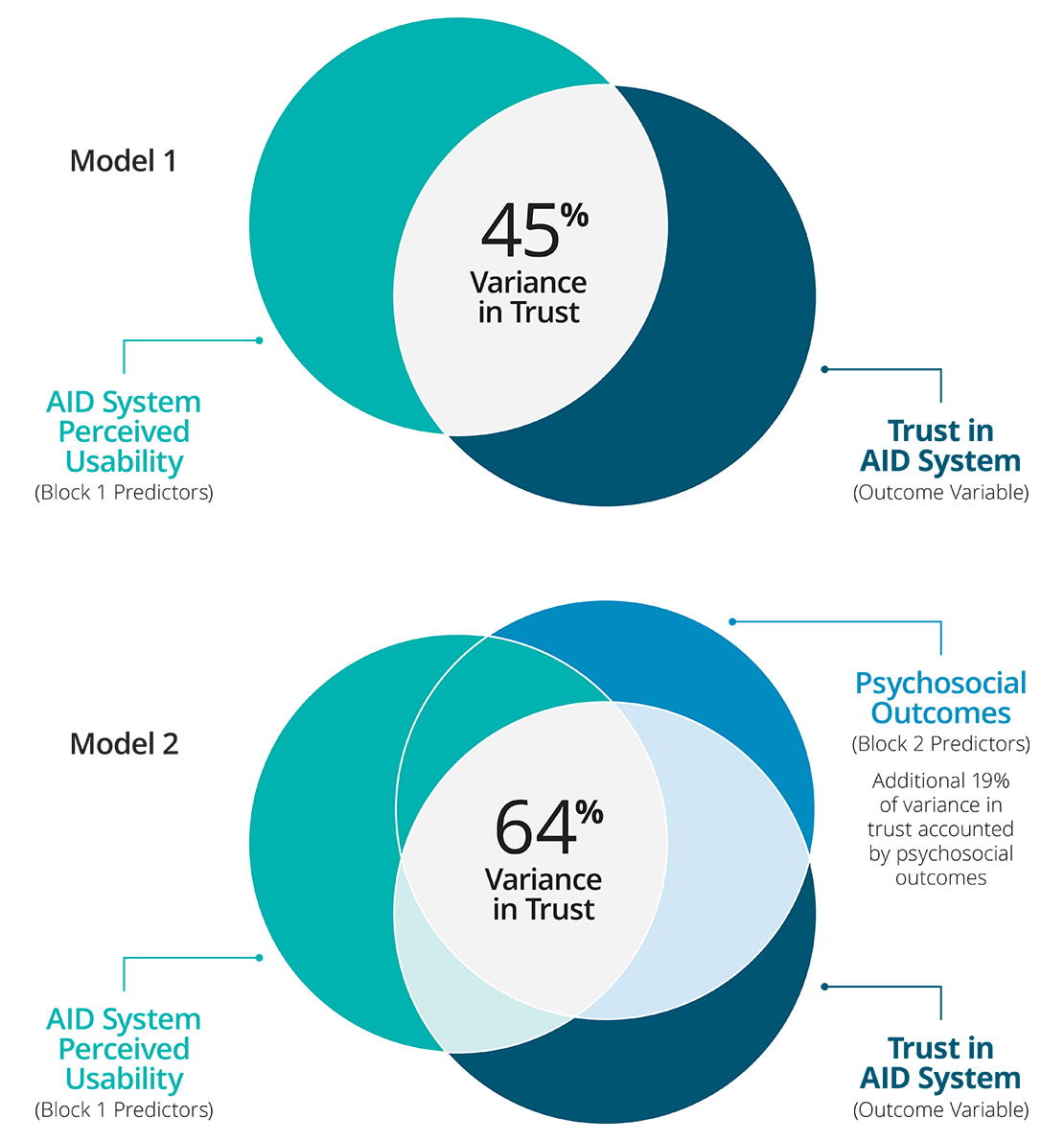
Hierarchical multiple regression analysis was used to ascertain the significance of ease of use (device level) and psychosocial (individual level) predictors of trust in AID systems. There were no issues with multicollinearity (all VIFs < 3). Ease of use accounted for 45% of variance in trust, F (3,382) = 106.74, p < .001. After psychosocial predictors were added variance accounted for was 64%, explaining an additional 19% of unique variance in trust, F (3,379) = 65.46, p < .001. Specifically the predictor variables (i.e., easy to use, too complicated, satisfaction, helps me have good BG control, and helps me sleep better at night) all significantly predict trust in AID systems.
Hierarchical Regression Results
Conclusion
Study findings suggest that both AID system perceived usability and psychosocial outcomes are distinctly informative and instrumental in the development of trust in AID systems.