Tandem Mobi System
Experience the tiny pump that’s big on control
What’s included in your free try-it-on kit

Wearability Like Never Before
The pump is so small that it can be worn almost anywhere,* giving you greater discretion, comfort, and options for how you manage your diabetes.
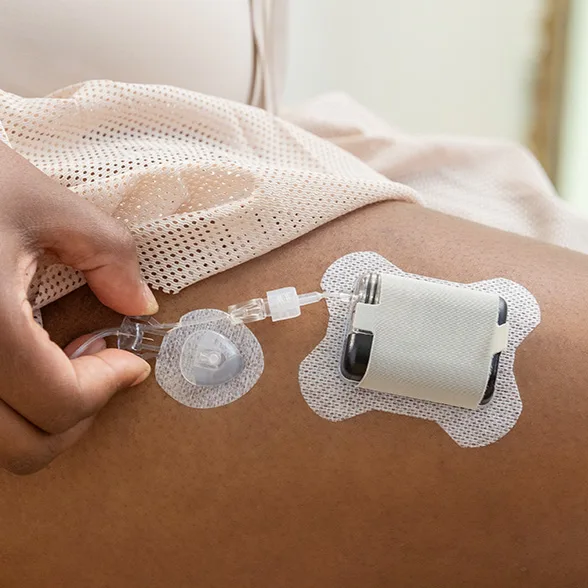
Wear it on-body with an adhesive sleeve,† which is compatible with all Tandem infusion sets.

Clip it easily to your clothes, dress, workout gear, or pajamas. Tandem Mobi can be worn almost anywhere.*

The impressively small Tandem Mobi is so discreet that you can slip it into the coin pocket of your jeans.
Disconnect. Reconnect.
Itʼs easy to temporarily disconnect an infusion set from your Tandem Mobi system for showering, hot tubs, or contact sports. Reconnecting is a snap!

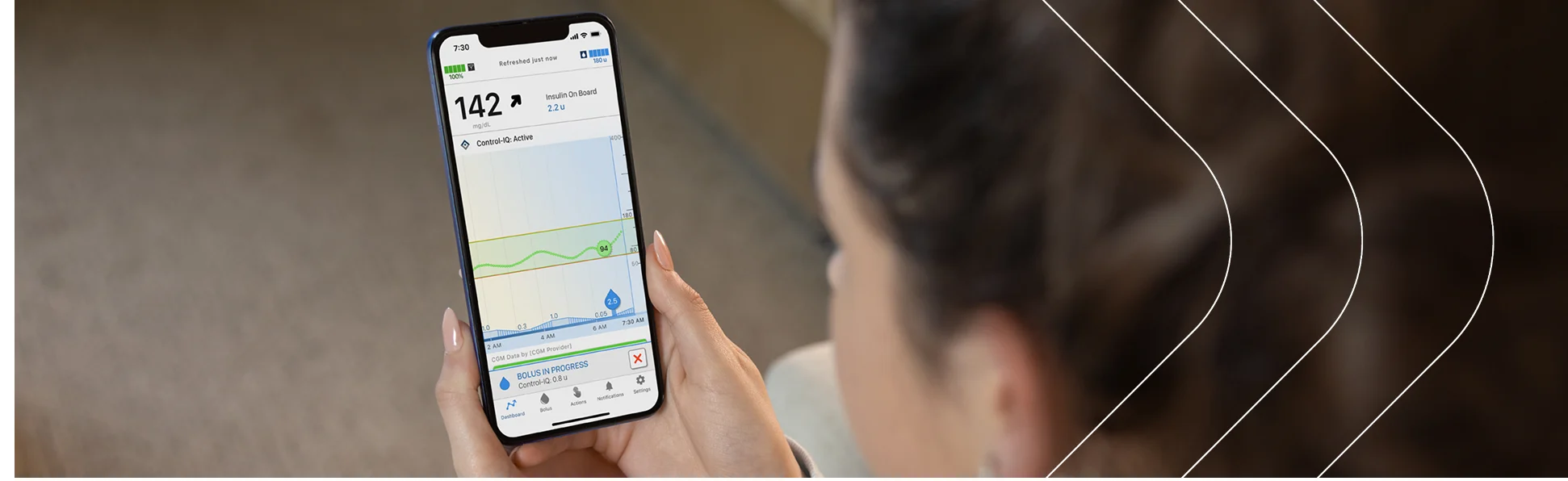
Tandem Mobi Mobile AppFree App Simulator
Test drive the easy-to-use interface of Tandem Mobi automated insulin delivery system with no obligation.
While your kit doesn’t include an infusion set, it would still be helpful to learn about the best ways to wear one. Plus, there are more than 35 mix-and-match infusion set options you can choose from.

*Do not wear or place your pump more than 12 inches (30.5 cm) above your infusion site. Doing so may result in over delivery of insulin.
† Adhesive sleeve sold separately.
Important Safety Information
RX ONLY.
Indications for Use
Tandem Mobi system: The Tandem Mobi insulin pump with interoperable technology (the pump) is intended for the subcutaneous delivery of insulin, at set and variable rates, for the management of diabetes mellitus in persons requiring insulin. The pump is able to reliably and securely communicate with compatible, digitally connected devices, including automated insulin dosing software, to receive, execute, and confirm commands from these devices. The pump is intended for single patient use. The pump is indicated for use in individuals 2 years of age and greater.
Control-IQ+ technology: Control-IQ+ technology is intended for use with compatible integrated continuous glucose monitors (iCGM, sold separately) and alternate controller enabled (ACE) pumps to automatically increase, decrease, and suspend delivery of basal insulin based on iCGM readings and predicted glucose values. It can also deliver correction boluses when the glucose value is predicted to exceed a predefined threshold. The Tandem Mobi system with Control-IQ+ technology is intended for the management of type 1 diabetes mellitus in persons 2 years of age and greater. Control-IQ+ technology is intended for single patient use. Control-IQ+ technology is indicated for use with NovoLog or Humalog U-100 insulin.
Warning: Control-IQ+ technology should not be used by anyone under the age of 2 years old. It should also not be used in patients who require less than 5 units of insulin per day or who weigh less than 20 pounds.
Control-IQ+ technology is not indicated for use in pregnant women, people on dialysis, or critically ill patients. Do not use Control-IQ+ technology if using hydroxyurea. Users of the Tandem Mobi pump and Control-IQ+ technology must: use the insulin pump, CGM, and all other system components in accordance with their respective instructions for use; test blood glucose levels as recommended by their healthcare provider; maintain sufficient diabetes self-care skills; see healthcare provider(s) regularly; and have adequate vision and/or hearing to recognize all functions of the pump, including alerts, alarms, and reminders. If your CGM readings do not match your symptoms or expectations, use a blood glucose meter to make diabetes treatment decisions. The Tandem Mobi pump, transmitter, and sensor must be removed before MRI, CT, or diathermy treatment. Exposure to MRI, CT, or diathermy treatment can damage the components. Visit tandemdiabetes.com/safetyinfo for additional important safety information.
These infusion sets are indicated for the subcutaneous infusion of insulin administered by Tandem insulin pumps for the treatment of diabetes. These infusion sets are indicated for single use. For contraindications, warnings, precautions, and other important information, please refer to the instructions for use accompanying the infusion sets.