Program Eligibility
If you qualify, and for a one-time payment, your new t:slim X2 insulin pump will remain under warranty with us for the duration of the previous pump’s warranty.
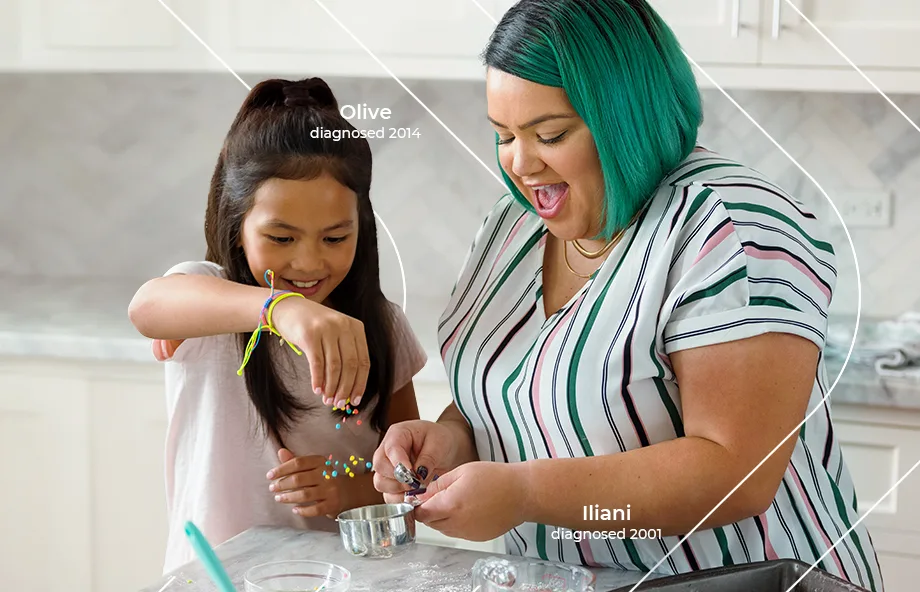
Our insulin pump with predictive technology is designed to offer a different approach to managing diabetes.
The Tandem Diabetes Care Pump Access Program allows non-Tandem pump users with in-warranty pumps to upgrade to the t:slim X2 insulin pump with Control-IQ technology for the remainder of their current pump warranty.

Helps Prevent Lows
Control-IQ technology predicts sensor glucose* 30 minutes ahead and decreases or stops basal insulin if glucose is predicted to be low.
Helps Prevent Highs
This powerful system can increase basal insulin if sensor glucose is predicted to be high, and is the only system that can deliver true automatic correction boluses.†
If you qualify, and for a one-time payment, your new t:slim X2 insulin pump will remain under warranty with us for the duration of the previous pump’s warranty.
One-time Payment | $1,399 |
|---|
Have at least six months of verifiable warranty remaining on your current insulin pump.
Cover the cost of supplies out‑of‑pocket or through your provincial program or private insurance.
Not be eligible for a new pump through your provincial program or private plan.
Fill out your information and a Tandem representative will reach out to answer your questions and help you sign up for the Pump Access Program
* CGM sold separately.
† If glucose values are
predicted to be above 10.0 mmol/l, Control-IQ technology calculates a correction bolus using the personal profile settings and a target of 6.1 mmol/l and delivers 60% of that value.
Terms and Conditions
Tandem Diabetes Care Canada, Inc. (“Tandem”) offers eligible participants a t:slim X2 insulin pump with Control-IQ technology (“Program Pump”) for an advance, one-time fee of $1,399 (4 years or less remaining in warranty). Eligible participants will be entitled to use the Program Pump for the remainder of the warranty of participant’s current non-Tandem insulin pump (the “Program Period”).
Supplies can be purchased through Diabetes Express.
At the end of the Program Period, the Program Pump must be returned to Tandem within 30 days in the same condition in which it was received by participant (less reasonable wear and tear), or participant shall purchase the pump for $6,300, less the applicable program pump price.
During the time participant is authorized by Tandem to possess the Program Pump, it will be covered by Tandem’s standard warranty.
Confirmation of participant’s current warranty will be required to start the program. Tandem reserves the right to deny participation in the program in its sole discretion. This offer may be changed or discontinued at any time and will expire on June 30, 2026.
Important Safety Information
This product may not be right for you. Always read and follow the label.
The t:slim X2 insulin pump with Control-IQ technology (the System) consists of the t:slim X2 insulin pump, which contains Control-IQ technology, and a compatible continuous glucose monitor (CGM, sold separately). The t:slim X2 insulin pump is intended for the subcutaneous delivery of insulin, at set and variable rates, for the management of diabetes mellitus in people requiring insulin. The t:slim X2 insulin pump can be used solely for continuous insulin delivery and as part of the System. When used with a compatible CGM, the System can be used to automatically increase, decrease, and suspend delivery of basal insulin based on CGM sensor readings and predicted glucose values. The System can also deliver correction boluses when the glucose value is predicted to exceed a predefined threshold. The pump and the System are indicated for use in individuals 6 years of age and greater. The pump and the System are intended for single user use. The pump and the System are indicated for use with NovoRapid, Admelog, Trurapi, or Humalog U-100 insulin. The System is intended for the management of Type 1 diabetes.
Warning: Control-IQ technology should not be used by anyone under the age of 6 years old. It should also not be used in users who require less than 10 units of insulin per day or who weigh less than 25 kilograms.
The System is not indicated for use in pregnant women, people on dialysis, or critically ill users. Do not use the System if using hydroxyurea.
Users of the pump and the System must: be willing and able to use the insulin pump, CGM, and all other system components in accordance with their respective instructions for use; test blood glucose levels as recommended by their healthcare provider; demonstrate adequate carb-counting skills; maintain sufficient diabetes self-care skills; see healthcare provider(s) regularly; and have adequate vision and/or hearing to recognize all functions of the pump, including alerts, alarms, and reminders. If your CGM readings do not match your symptoms or expectations, use a blood glucose meter to make diabetes treatment decisions. The t:slim X2 pump must be removed before MRI, CT, or diathermy treatment. Visit tandemdiabetes.com/safetyinfo for additional important safety information.